Autoimmune Brain Panel™
Autoantibody testing for complex neuropsychiatric presentations
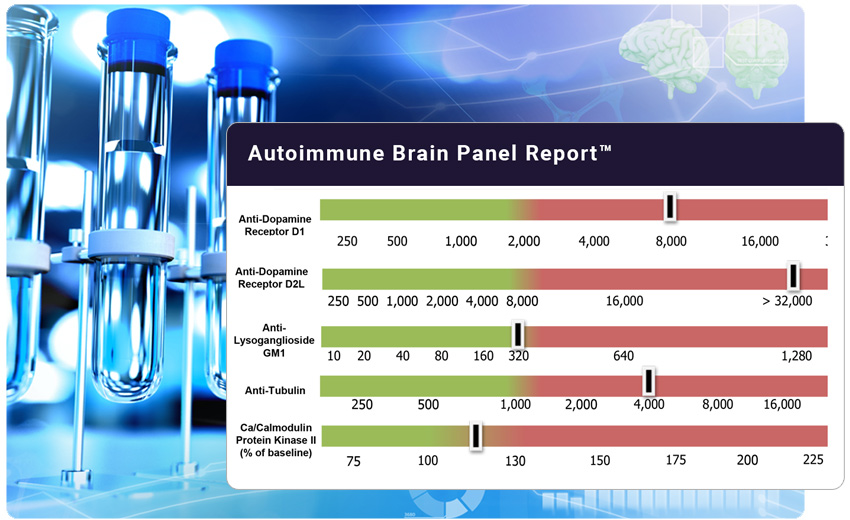
The Autoimmune Brain Panel™ (formerly the Cunningham Panel™) includes a series of five clinically validated assays that help identify whether a patient’s neuropsychiatric symptoms may be due to an underlying autoimmune process, often triggered by infection.
The Panel measures the levels of circulating autoantibodies in serum that are directed against Dopamine D1 and D2 receptors, Lysoganglioside GM-1 and Tubulin. The fifth test, the CaMKII, is a cell stimulation assay. This test measures the ability of the patient’s autoantibodies to stimulate the CaMKII enzyme that is responsible for the upregulation of brain neurotransmitters.
Using Results to Guide Patient Care
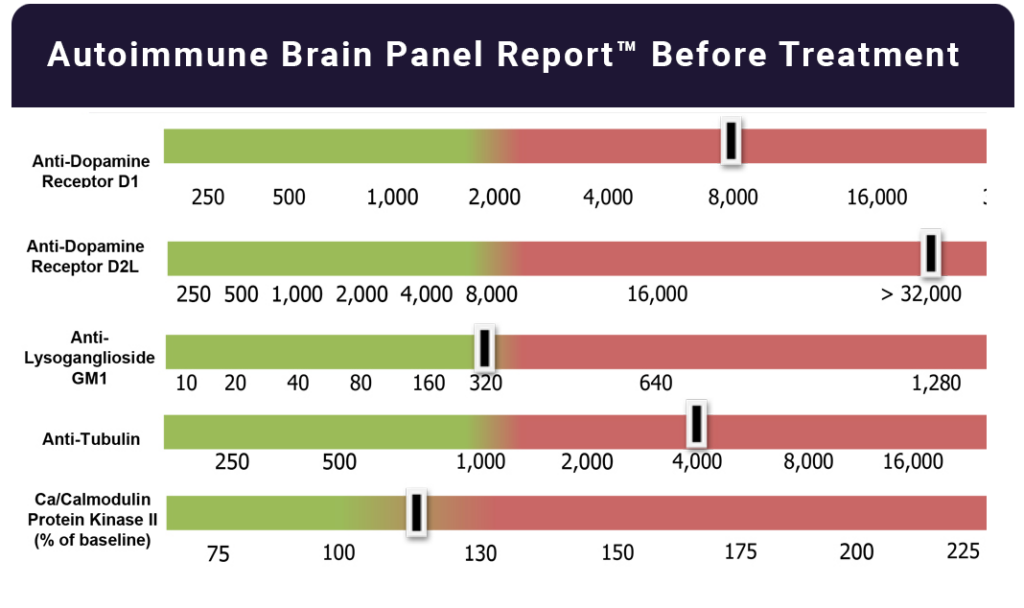
The Autoimmune Brain Panel™ can help clinicians determine whether a patient’s neuropsychiatric symptoms may be associated with an infection-triggered autoimmune response rather than a primary neurologic or psychiatric disorder.
Identifying an autoimmune component can be important, as it may influence treatment strategies, including consideration of immunomodulatory and antimicrobial approaches, rather than relying solely on psychotropic medications.
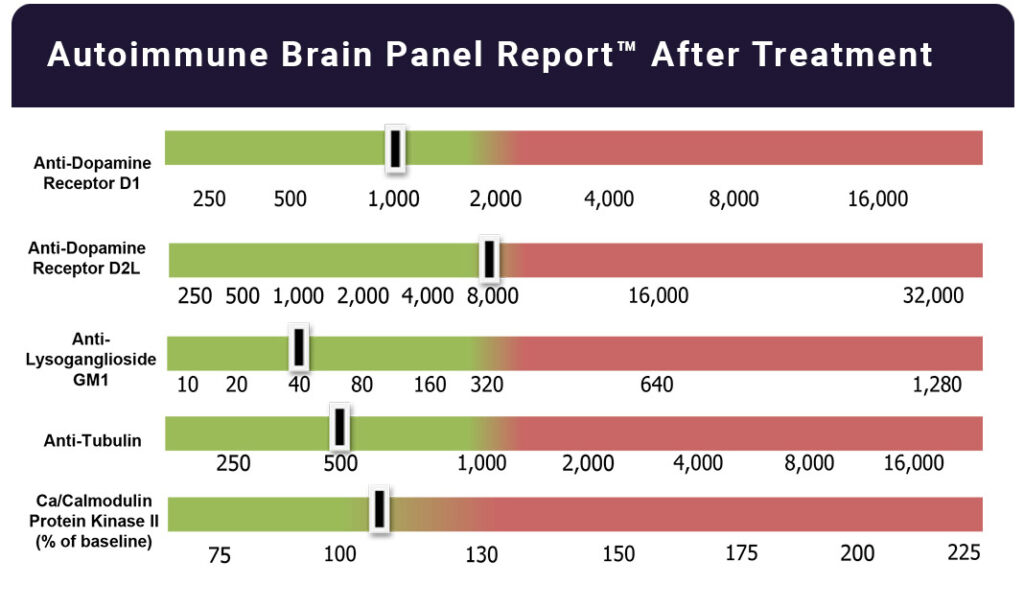
Once these conditions are properly identified and treated, many patients report dramatic improvement — ranging from significant symptom reduction to complete resolution.
Neuropsychiatric Symptoms Associated with Neuronal Autoantibody Targets
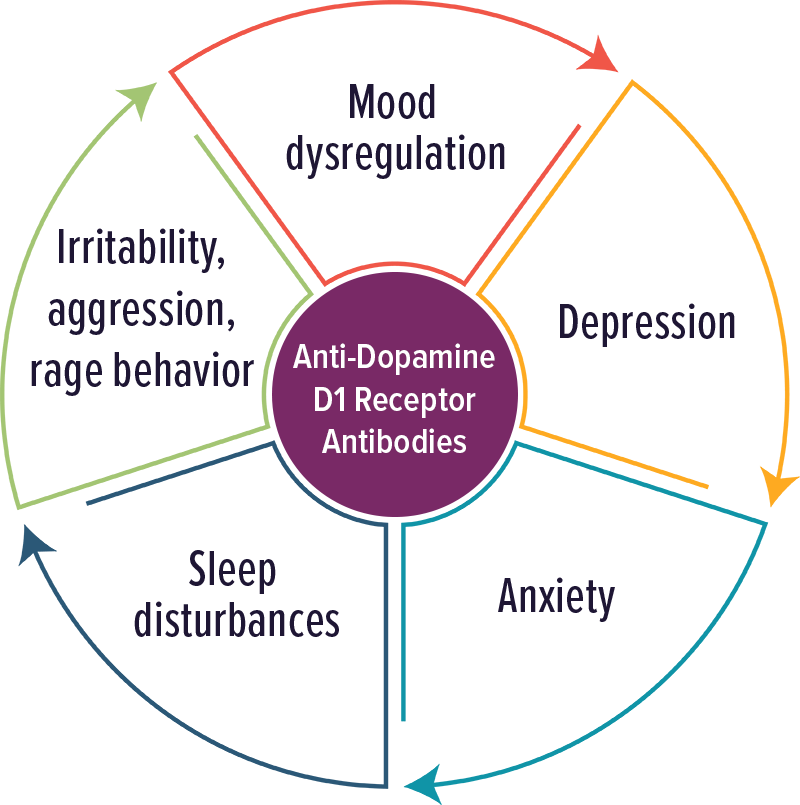
Dopamine D1 Receptor Antibodies
Individuals with elevated levels of autoantibodies against Dopamine D1 receptor typically experienced psychiatric symptoms, including psychosis. Other symptoms included: mood dysregulation, anxiety, depression, sleep disturbances, irritability, aggression and rage behavior.
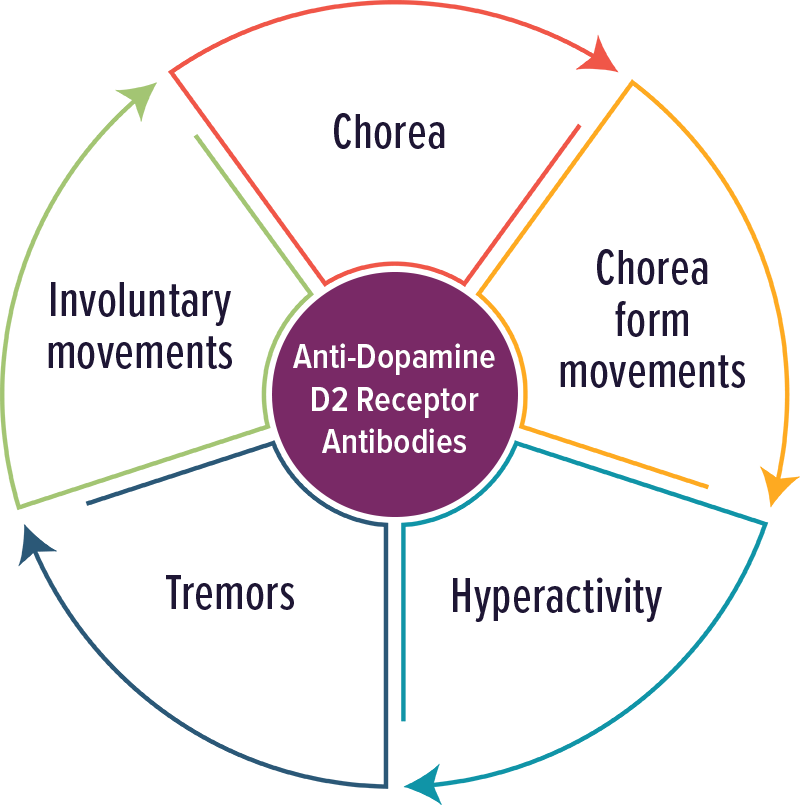
Dopamine D2 Receptor Antibodies
Individuals with elevated levels of autoantibodies against Dopamine D2 receptor typically experienced movement disorders and impulsivity. Other symptoms included: chorea, chorea form movements, hyperactivity, tremors and involuntary movements.
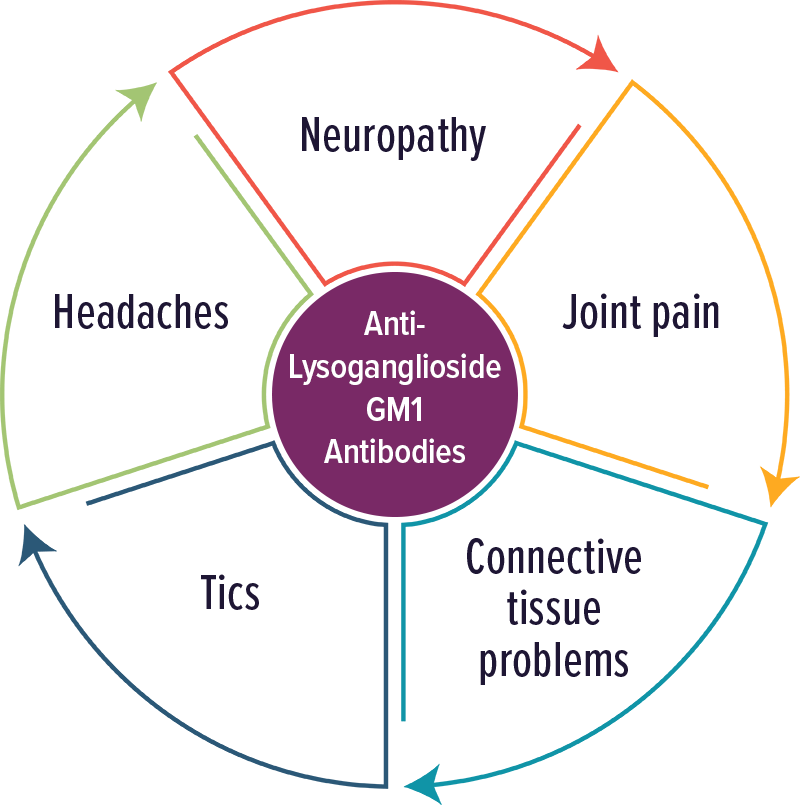
Lysoganglioside GM1 Antibodies
Individuals with elevated levels of autoantibodies against Lysoganglioside GM1 typically experienced neuropathic symptoms, including tics. Other symptoms included: neuropathy, joint pain, connective tissue problems, tics and headaches.
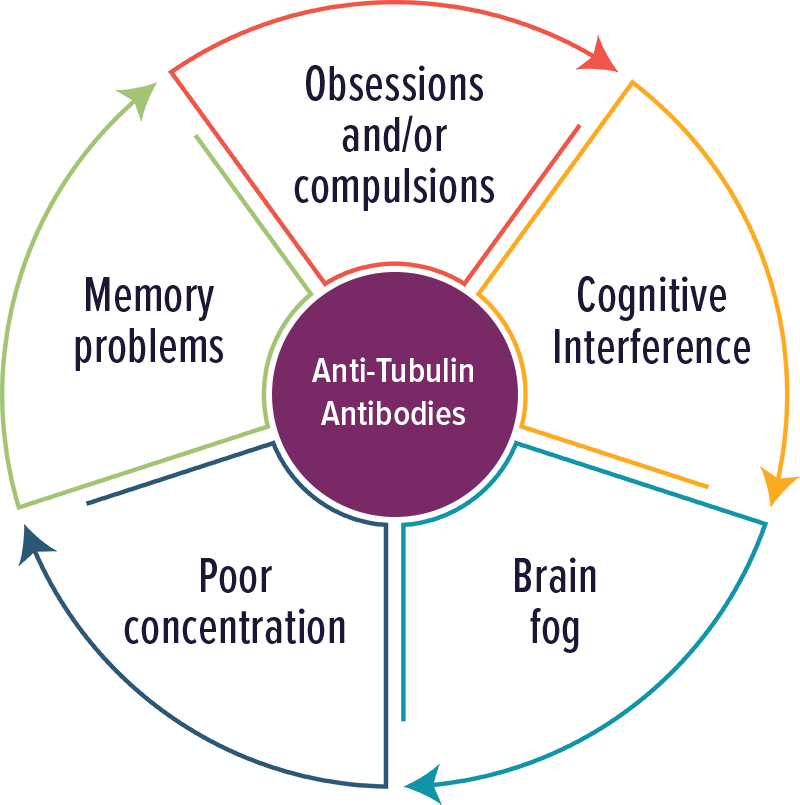
Tubulin Antibodies
Individuals with elevated levels of autoantibodies against Tubulin typically experienced cognitive complaints, OCD and brain fog. Other symptoms included: poor concentration and memory problems.
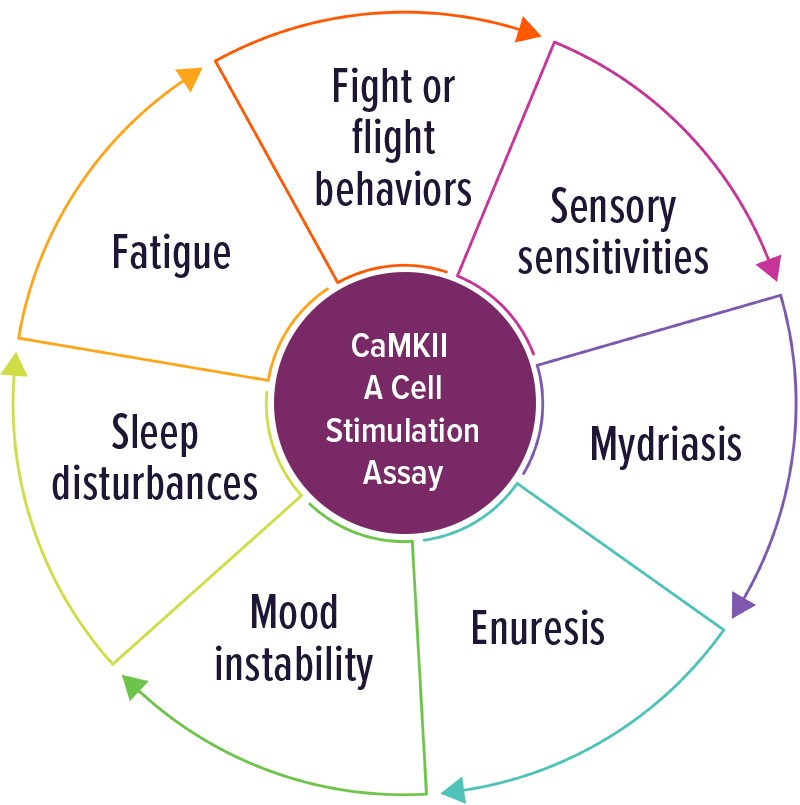
CaMKII – A Cell Stimulation Assay
Individuals with elevated CaMKII levels were often positive with involuntary movements and any symptom of adrenergic activation. Other symptoms included: fight or flight behaviors, sensory abnormalities, fatigue, sleep disturbance, mood instability, enuresis and mydriasis.
References
- Shimasaki C, Frye RE, Trifiletti R, Cooperstock M, Kaplan G, Melamed I, Greenberg R, Katz A, Fier E, Kem D, Traver D, Dempsey T, Latimer ME, Cross A, Dunn JP, Bentley R, Alvarez K, Reim S, Appleman J. Evaluation of the Cunningham Panel™ in pediatric autoimmune neuropsychiatric disorder associated with streptococcal infection (PANDAS) and pediatric acute-onset neuropsychiatric syndrome (PANS): Changes in antineuronal antibody titers parallel changes in patient symptoms. J Neuroimmunol. 2020 Feb 15;339:577138. doi: 10.1016/j.jneuroim.2019.577138. Epub 2019 Dec 15. PMID: 31884258. https://www.jni-journal.com/article/S0165-5728(19)30352-2/fulltext
Test Sensitivity, Specificity, and Accuracy
Study findings demonstrated a strong positive association between changes in neuropsychiatric symptoms and changes in anti-neuronal autoantibody levels and antibody-mediated CaMKII activation in human neuronal cells.
Based on changes in the number of positive assays, the overall accuracy was 86%, with sensitivity of 88% and specificity of 83%.
The study results suggest there may be clinical utility in monitoring autoantibody levels and CaMKII stimulatory activity against these five neuronal targets when diagnosing and treating patients with immune-mediated neuropsychiatric disorders.
Results you can count on.
Clinical Utility Supported by Multiple Studies
The clinical utility of the Autoimmune Brain Panel™ is supported by multiple peer-reviewed studies evaluating the role of anti-neuronal autoantibodies and CaMKII activation in patients with neuropsychiatric symptoms.
These studies demonstrate associations between autoantibody activity, symptom severity, and clinical response to treatment, supporting the Panel’s use as a tool to help clinicians evaluate potential immune-mediated contributions to neuropsychiatric disorders.
Consultation for Utilization of the Panel
We provide free, medical education services to assist clinicians in better understanding the Autoimmune Brain Panel™ results. Please fill out the form below and we will contact you to set up a convenient time for you. Note: This service is open to healthcare providers only.