Autoimmune Brain Panel™
A clinical tool to help evaluate possible autoimmune causes of neuropsychiatric symptoms.
Designed for patients with sudden-onset or treatment-resistant neuropsychiatric symptoms.
The Autoimmune Brain Panel™ (formerly the Cunningham Panel™) is a set of five clinically validated assays that help identify whether a patient’s neurologic or psychiatric symptoms may have an underlying autoimmune component.
The panel measures four autoantibodies—targeting dopamine D1 and D2 receptors, lysoganglioside GM1, and tubulin—that can disrupt neuronal signaling, drive neuroinflammation, and contribute to symptoms such as anxiety, OCD, tics, mood changes, and cognitive dysfunction.
Go Page Below
Go Page Below

What Does the Autoimmune Brain Panel™ Measure?
The Autoimmune Brain Panel™ is a series of five proprietary, validated tests. Four of these tests measure circulating levels of specific autoantibodies in a patient’s serum at the time the specimen is drawn.
The fifth test, the CaMKII assay, measures the ability of a patient’s autoantibodies to stimulate this enzyme, resulting in an upregulation (or increase) of brain neurotransmitters such as dopamine, epinephrine and norepinephrine. This increase can trigger a variety of neurologic and/or psychiatric symptoms.
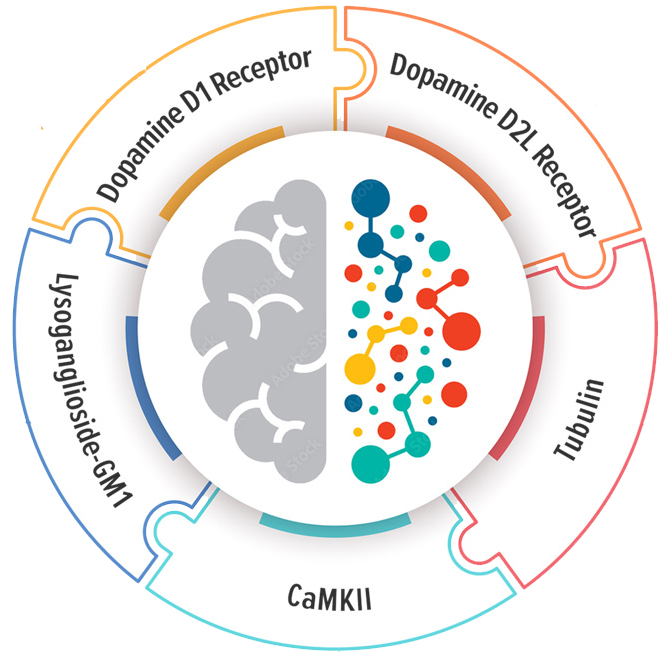
Autoimmune Brain Panel™ Includes 5 Individual Tests
The neuronal targets included in the Autoimmune Brain Panel™ were selected based upon their biological association with specific neurologic and psychiatric symptoms.
Background on Dopamine Receptors
The dopamine receptors (D1, D2, D3, D4, D5) are widely distributed in the brain and mediate the effects of dopamine on: cognition, emotion, regulation of hunger, satiety, locomotor activity and on the endocrine system. 1 Dopamine receptors are highly concentrated on synaptic neurons in the brain.
The dysregulation of the dopaminergic system has been linked to the pathophysiology of many diseases, such as schizophrenia, attention deficit/hyperactivity disorder, and depression. 1
Autoantibodies against Dopamine D1 Receptor
When a patient’s autoantibodies are directed against this receptor, they can interfere with the normal function of this receptor by stimulating the receptor or by blocking the ability of dopamine to bind to this receptor.
This disruption of dopamine transmission can lead to the manifestation of various neuropsychiatric disorders. 2, 3
Individuals with elevated levels of anti-Dopamine D1 antibodies often reported having psychiatric symptoms including psychosis, OCD behaviors and tics, based upon our clinical laboratory patient population analysis.
Background on Dopamine Receptors
The dopamine receptors (D1, D2, D3, D4, D5) are widely distributed in the brain and mediate the effects of dopamine on: cognition, emotion, regulation of hunger, satiety, locomotor activity and on the endocrine system. 1 Dopamine receptors are highly concentrated on synaptic neurons in the brain.
The dysregulation of the dopaminergic system has been linked to the pathophysiology of many diseases, such as Alzheimer’s disease, schizophrenia, Parkinson’s disease, attention deficit/hyperactivity disorder, depression and drug addiction. 1
Autoantibodies against Dopamine D2 Receptor
Autoantibodies directed against this receptor can interfere with its normal function through overstimulation or by blocking dopamine’s normal binding with the receptor.
Autoantibodies directed against D2 receptors correlated with various neuropsychiatric symptoms. 4
Individuals with elevated levels of anti-Dopamine D2 antibodies often reported having symptoms involving uncontrolled motor movements, such as hyperactivity and impulsivity, based on analysis of our clinical laboratory population.
Autoantibodies Against Lysoganglioside-GM1
Lysoganglioside GM1 is located within the membrane of nerve cells and is highly concentrated in the central nervous system. It functions as insulation around the nerve cell and plays an important role in signal transmission in the brain. It provides a vital function in communication between neurons and ensures proper transmission of impulses.
When a patient’s autoantibodies are directed against lysoganglioside, they can interrupt this communication and interfere with normal nerve transmission.
Individuals with elevated levels of anti-Lysoganglioside GM1 often reported having joint pain, connective tissue pain, headaches, and other types of neuropathy, based upon our clinical laboratory patient population analysis.
An autoimmune response against ganglioside GM1 has been implicated in Guillain-Barré Syndrome. 4,5,6
Autoantibodies Against Tubulin
Tubulin is an intracellular protein that forms microtubules providing a skeleton for maintaining cell shape and is thought to be involved in cell motility and intracellular transport.
Tubulin is contained within most every cell and is highly abundant and concentrated in brain cells. It plays an important role in cell signaling and communication within the cell.
When a patient’s autoantibodies are directed against Tubulin, OCD-like symptoms and cognitive impairment, such as ‘brain fog’ have been reported, based upon our clinical laboratory patient population analysis.
CaMKII – a Cell Stimulation Assay
Calcium/calmodulin-dependent protein kinase II (CaMKII) is a key enzyme that is involved in the upregulation of neurotransmitters: dopamine, epinephrine and norepinephrine.
CaMKII is involved in regulating various neuronal functions, such as neurotransmitter synthesis and release, receptor signaling, and long-term synaptic plasticity. It plays a crucial role in synaptic transmission and transmitter release.
The CaMKII test performed in the Autoimmune Brain Panel™ is a “cell stimulation assay.” This test involves growing human brain cells in culture and incubating the patient’s serum on these cells to determine if autoantibodies that are present bind to and stimulate this enzyme.
If a patient’s autoantibodies stimulate this enzyme, it can trigger abnormal neurologic, psychiatric and behavioral symptoms. 7.
Elevated levels of CaMKII tend to indicate the presence of active infection of some type.
Interpreting Test Results
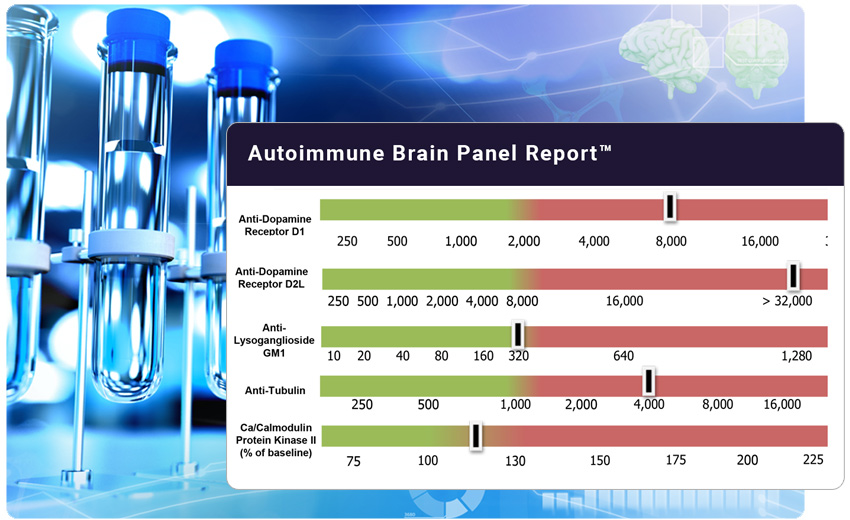
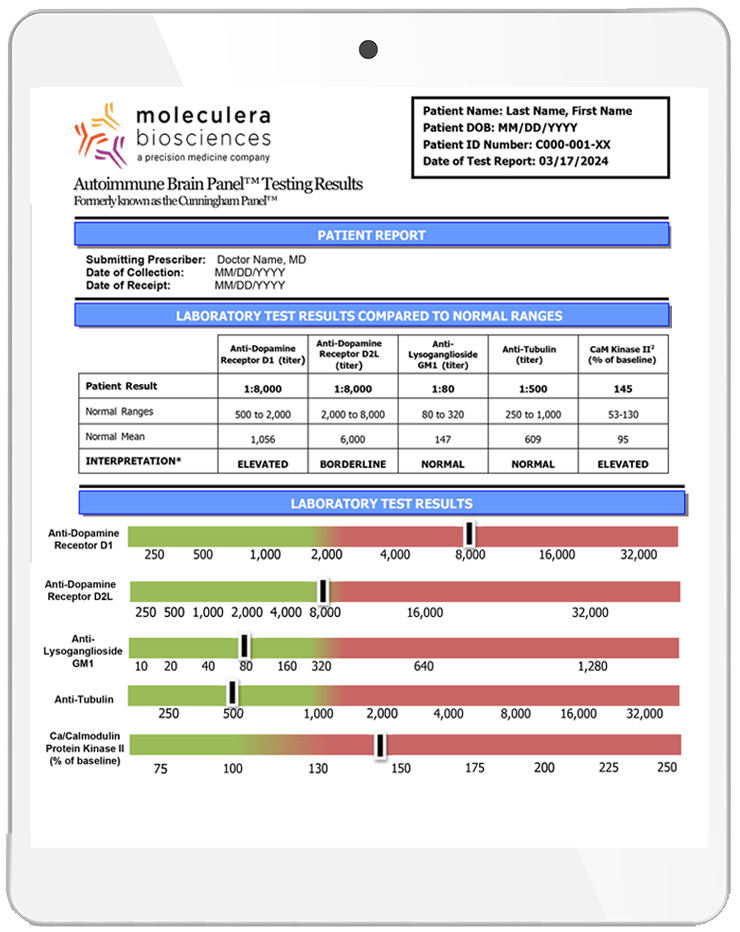
The Autoimmune Brain Panel™ is considered positive if one or more of these individual test results exceed their normal ranges. Elevated levels may indicate that neurologic or psychiatric symptoms are associated with an underlying autoimmune process.
The autoantibody test results are expressed as titers (or final dilution) at which an endpoint was observed on an Enzyme-Linked Immunosorbent Assay (ELISA) format.
The CaMKII is a cell stimulation assay, which measures the stimulatory ability of a patient’s autoantibody IgGs to increase the activity of the CaMKII enzyme within a human brain cell line. The result is a numeric score that reflects the percent above or below baseline activity.
Test Sensitivity, Specificity, and Accuracy
Study findings demonstrated a strong positive association between changes in neuropsychiatric symptoms and changes in anti-neuronal autoantibody levels and antibody-mediated CaMKII activation in human neuronal cells.
Based on changes in the number of positive assays, the overall accuracy was 86%, with sensitivity of 88% and specificity of 83%.
The study results suggest there may be clinical utility in monitoring autoantibody levels and CaMKII stimulatory activity against these five neuronal targets when diagnosing and treating patients with immune-mediated neuropsychiatric disorders.
Results you can count on.
Clinical Utility Supported by Multiple Studies
The clinical utility of the Autoimmune Brain Panel™ is supported by multiple peer-reviewed studies evaluating the role of anti-neuronal autoantibodies and CaMKII activation in patients with neuropsychiatric symptoms.
These studies demonstrate associations between autoantibody activity, symptom severity, and clinical response to treatment, supporting the Panel’s use as a tool to help clinicians evaluate potential immune-mediated contributions to neuropsychiatric disorders.
Go Page Below
The Autoimmune Brain Panel™ is designed to help uncover whether neuropsychiatric symptoms are being driven by an infection-triggered autoimmune response rather than a primary psychiatric or neurological disorder.
Identifying an autoimmune component is important, since it typically changes the course of treatment to focus on the underlying infections and immune dysfunction, rather than solely treating with psychotropic medications.
Once these conditions are properly identified and treated, many patients report dramatic improvement — ranging from significant symptom reduction to complete resolution.
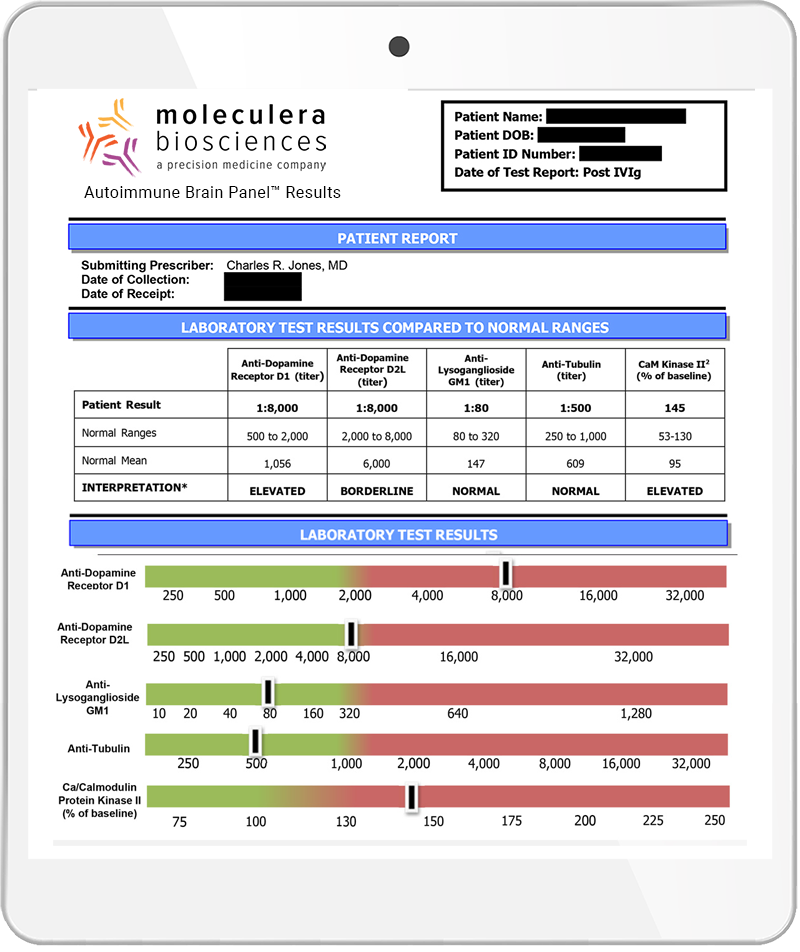
Neuropsychiatric Symptoms and Test Biomarkers
Anti-Dopamine D1 Receptor Antibodies
Individuals with elevated levels of autoantibodies against Dopamine D1 receptor typically experienced psychiatric symptoms, including psychosis.
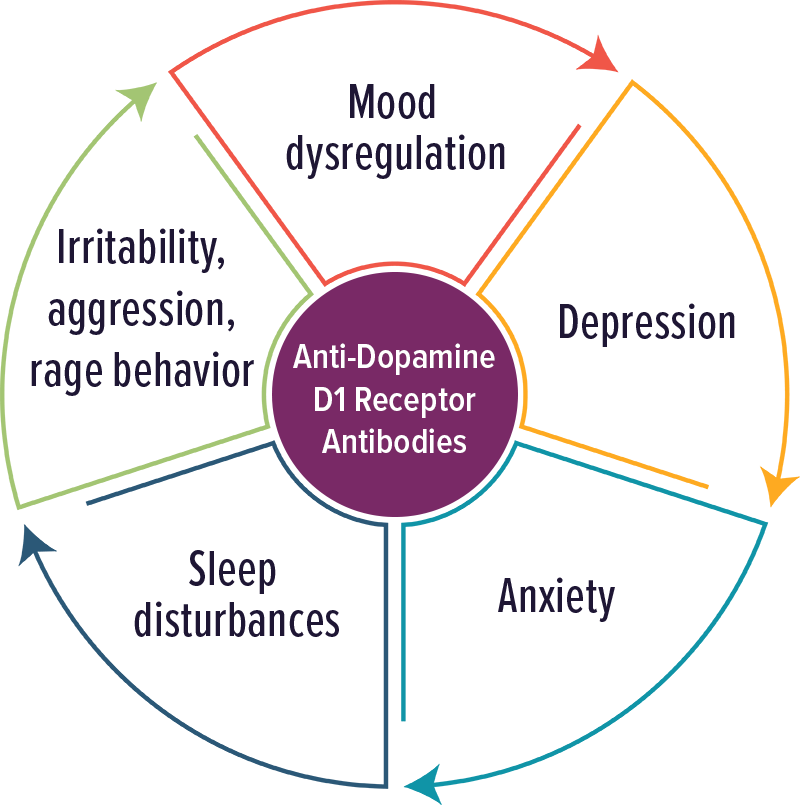
Anti-Dopamine D2 Receptor Antibodies
Individuals with elevated levels of autoantibodies against Dopamine D2 receptor typically experienced movement disorders and impulsivity.
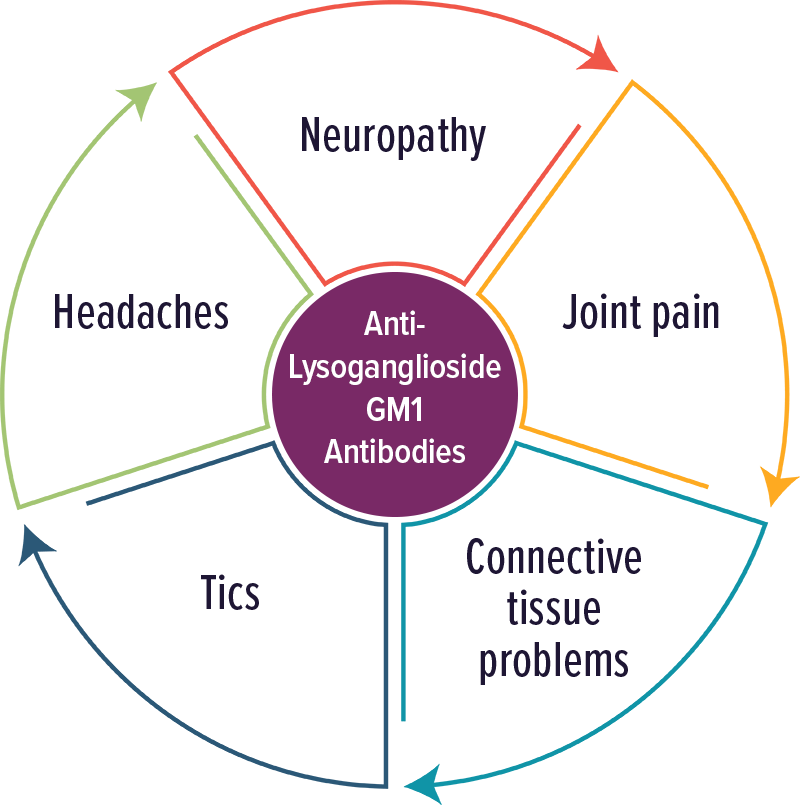
Anti-Lysoganglioside GM1 Antibodies
Individuals with elevated levels of autoantibodies against Lysoganglioside GM1 typically experienced neuropathic symptoms, including tics.
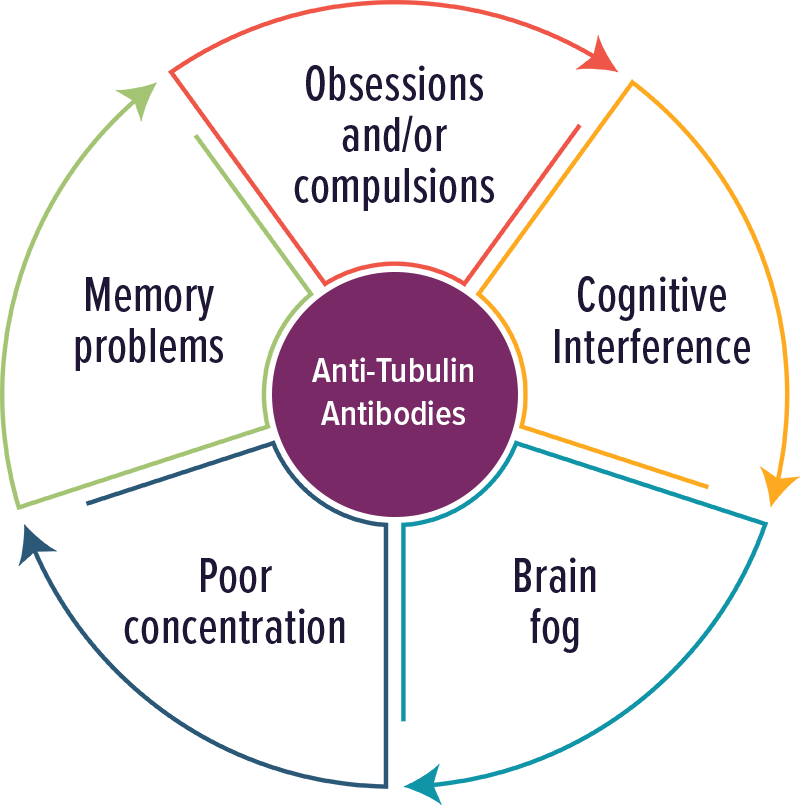
Anti-Tubulin Antibodies
Individuals with elevated levels of autoantibodies against Tubulin typically experienced cognitive complaints, OCD and brain fog.
CaMKII – A Cell Stimulation Assay
Individuals with elevated CaMKII levels were often positive with involuntary movements and any symptom of adrenergic activation.
An elevated CaMKII result indicates a patient’s autoantibodies are stimulating this enzyme. CaMKII is responsible for upregulating brain neurotransmitters such as dopamine, epinephrine and norepinephrine.
CaMKII activation may result in disrupted neuronal functioning, and trigger a variety of neurologic and/or psychiatric symptoms.